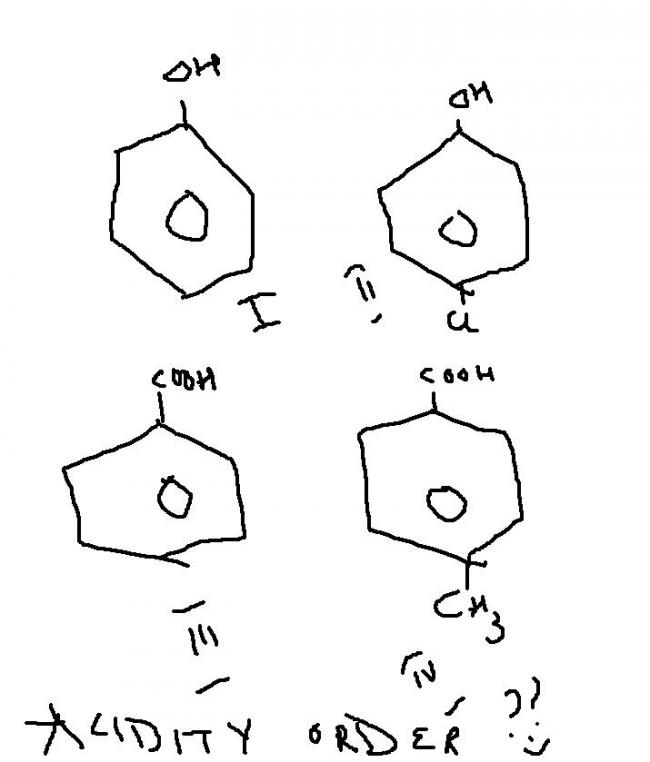
ok..here goes d reason..
2 is d correct options...coz for acidity we chek for stability of conjuagte base..
so after H is removed..
3 n 4 give equivalent canonical forms...which is highly stable....so 3,4 >1,2
now 4 has a methyl wid a +I effect from meta position which whill destabilise d carboanion,,,
so 3>4>2>1,,,
1 is a normal form..while 2 has -I effect that will help stabilise d carboanions...
hence..B is d answer...