8 Which of the following common names does not represent a dicarboxylic acid?
A) lactic acid
B) succinic acid
C) phthalic acid
D) glutaric acid
--------------------------------------------------------------------------------
9 Which of the following statements is not generally true?
A) the boiling point of a carboxylic acid is higher than that of its methyl ester.
B) methyl esters are more reactive acylating agents than their amide counterparts.
C) amide hydrolysis may be carried out with either strong acid or base catalysis.
D) Fischer esterification of acids with alcohols requires a strong base catalyst.
--------------------------------------------------------------------------------
10 What is the order of increasing acidity for the following compounds? (weaker < stronger) I 4-methylpentanoic acid II 3-chloropentanoic acid III 2-bromopentanoic acid IV 2,2-dichloropentanoic acid
A) I < II < III < IV
B) IV < III < II < I
C) I < III < II < IV
D) II < III < I < IV
--------------------------------------------------------------------------------
11 When comparing the acidity of propanoic acid and pyruvic acid, CH3COCO2H, which of the following statements is correct.
A) propanoic acid has a lower pKa and a smaller Ka than pyruvic acid
B) propanoic acid has a lower pKa and a larger Ka than pyruvic acid
C) propanoic acid has a higher pKa and a larger Ka than pyruvic acid
D) propanoic acid has a higher pKa and a smaller Ka than pyruvic acid
--------------------------------------------------------------------------------
12 Consider the following dicarboxylic acids? I adipic acid
HO2C(CH2)4CO2H II malonic acid
CH2(CO2H)2 III oxalic acid
(CO2H)2 IV succinic acid
HO2C(CH2)2CO2H
What is the order of increasing acid strength? (weaker < stronger)
A) I < II < III < IV
B) IV < III < II < I
C) I < IV < II < III
D) II < I < IV < III
--------------------------------------------------------------------------------
13 Dicarboxylic acids have two pKa's.
For maleic acid (cis-2-butenedioic acid) these are pKa1 = 2.0, and pKa2 = 6.3
For fumaric acid (trans-2-butenedioic acid) these are pKa1 = 3.0, and pKa2 = 4.5
Which factor best explains why the cis-isomer has a smaller pKa1 and a larger pKa2 than the trans-isomer?
A) intramolecular steric hindrance
B) intramolecular dipole repulsion
C) intramolecular hydrogen bonding
D) selective solvation in water
--------------------------------------------------------------------------------
14 An equimolar mixture of benzoic acid and benzyl alcohol is dissolved in equal volumes of ether and 5% aqueous NaOH.
The resulting mixture separates into two immiscible liquid layers. Which of the following is approximately correct?
A) both organic solutes are largely in the ether layer
B) the benzyl alcohol is in the ether layer and the benzoic acid is in the water layer
C) both organic solutes are largely in the water layer
D) the benzyl alcohol is in the water layer and the benzoic acid is in the ether layer
--------------------------------------------------------------------------------
15 Fischer esterification of phenylacetic acid with 1-propanol gave a mixture of 93% of the ester, propyl phenylacetate, contaminated with 7% unreacted acid.
Which of the following treatments would be best used to purify the ester?
A) reduce the unwanted acid with LiAlH4 in ether.
B) wash an ether solution of the crude product with concentrated brine (aq. NaCl).
C) wash an ether solution of the crude product with 5% aqueous sulfuric acid.
D) wash an ether solution of the crude product with 5% aqueous sodium carbonate.
--------------------------------------------------------------------------------
16 Which of the following would not be a useful reaction for preparing isobutyric acid, (CH3)2CHCO2H?
A) 2-methyl-1-propanol + Jones' reagent
B) 2-bromopropane + CO2; followed by hydrolysis
C) cis-2,5-dimethyl-2-hexene + O3; followed by H2O2
D) 2-bromopropane + NaCN; followed by acid-catalyzed hydrolysis
--------------------------------------------------------------------------------
17 Which of the following would not be a useful method for converting a carboxylic acid into an ester derivative?
A) RCO2H + CH2N2 in ether
B) RCO2H + (CH3)2C=CH2 & acid catalyst
C) RCO2H + C2H5OH & acid catalyst + heat (-H2O)
D) RCO2(-) Na(+) + (CH3)3CBr
--------------------------------------------------------------------------------
18 Which of the following is an intermediate in the Fischer esterification of propanoic acid with ethanol?
A) 1-propoxy-1,1-dihydroxyethane
B) 2-propoxy-1,1-dihydroxyethane
C) 1-ethoxy-1,1-dihydroxypropane
D) 2-ethoxy-1,1-dihydroxypropane
--------------------------------------------------------------------------------
19 Which of the following reagents does not react with benzoic acid, converting it into a different compound?
A) NaI in acetone
B) SOCl2
C) LiAlH4 in ether
D) excess CH3Li in pentane
--------------------------------------------------------------------------------
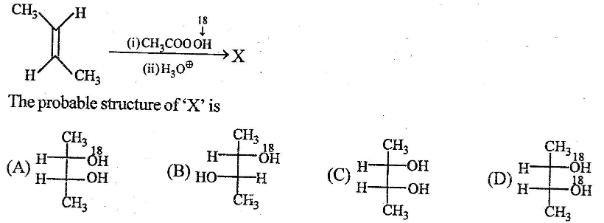
20 Which statement regarding isotope exchange of 18OH2 with the oxygen of a carboxyl group is correct?
no exchange occurs under any circumstances
base-catalyzed exchange is more effective than acid-catalyzed exchange
acid-catalyzed exchange is more effective than base-catalyzed exchange
acids and bases are equally effective in catalyzing an exchange
--------------------------------------------------------------------------------
21 Which of the following compounds could not be converted into pivalic acid ( 2,2-dimethylethanoic acid ) in three or fewer steps?
A) 2,2-dimethyl-1-butene
B) 2,3-dimethyl-2-butene
C) 2,2-dimethyl-1-propanol
D) 2-bromo-2-methylpropane
--------------------------------------------------------------------------------
22 Treatment of benzoic acid with excess 18OH2 and a strong acid catalyst results in what change?
A) both oxygens of the carboxyl group exchange with the 18O of the water
B) only the carbonyl oxygen exchanges with the 18O of the water
C) only the OH oxygen exchanges with the 18O of the water
D) no change takes place
--------------------------------------------------------------------------------
23 Two C8H9Br isomers form Grignard reagents which on carboxylation give isomeric carboxylic acids.
Oxidation of each isomeric acid with hot KMnO4 produces the same C9H6O6 tricarboxylic acid.
Which of the following compounds are the original isomeric bromides?
A) I & II
B) III & IV
C) I & IV
D) II & III
--------------------------------------------------------------------------------
24 Which of the following reagents will reduce a carboxylic acid to a 1 º-alcohol under mild conditions? I BH3 in ether II NaBH4 in ethanol III H2 & Pt catalyst IV LiAlH4 in ether
A) I & IV
B) II & III
C) only III
D) all of them
--------------------------------------------------------------------------------
25 What is the order of increasing base strength for the following salts? (weaker < stronger) I sodium ethoxide II potassium formate III sodium benzoate IV sodium dichloroacetate
A) I < II < III < IV
B) IV < II < III < I
C) II < III < IV < I
D) IV < I < II < III